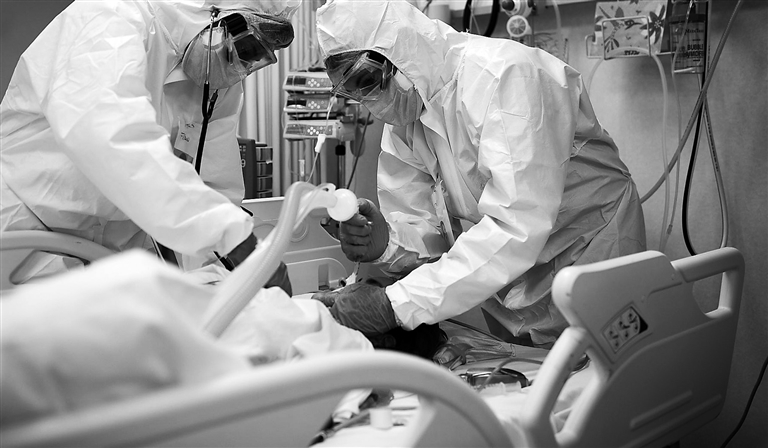
AN Israeli public company says it developed the “only cure for late-stage COVID” and said that the results of its phase II clinical trial revealed that patients suffering from severe cases of COVID-19 had a 94 percent survival rate after being treated with the drug. Israeli biotechnology company Bonus BioGroup’s cell therapy MesenCure was administered to 50 hospitalized COVID-19 patients suffering from life-threatening pneumonia and respiratory distress, the company said, noting that 47 of those patients had survived. “These are the most clinically meaningful results presented today for treating severe COVID-19 patients,” Dr. Tomer Bronshtein, the head of research at Bonus BioGroup Ltd., told Fox News in an interview. Bronshtein said the phase II clinical trial, which recently concluded and took place in several medical centers in Israel, revealed that MesenCure can “save three out of four people that are at risk of dying” and “can reduce their length of hospital stay by half.” He said it “will accelerate their healing in a way that will cause less permanent tissue damage and reduce the risk for ‘long COVID.’” The company noted that for each of the first 30 participants in the clinical trial, two patients, also suffering from severe cases, with similar ages and comorbidities, were selected to the “control group,” which did not receive MesenCure. After analyzing the data, the company found that Mesencure reduced about 70 percent of severe COVID-19 patients’ mortality compared to the mortality in the group that did not receive the drug. The company also noted that about half of the patients with severe cases of COVID-19 who were treated with the therapy were discharged from the hospital up to just one day after the end of the treatment. “MesenCure is a drug made from living cells,” Bronshtein explained, noting that the cells “are infused into the patient” and once the cells “reach their lungs, they fill the inflammation and start secreting molecules and compounds that soothe this inflammation.” Bronshtein noted that it also reduces pneumonia and edema, which helps patients “be able to breathe.” “In parallel, [the cells] also ameliorate what’s called the cytokine storm, which is the hyperimmune reaction that is aggravated in COVID 19,” he added. Bonus BioGroup argued that it is “necessary to provide a therapeutic option for hospitalized, severe COVID-19 patients” given “vaccines reduce, but do not entirely prevent COVID-19 infection and severe illness, which is also the case with medications designed for at-home treatment of mild COVID-19 that may reduce, but not prevent hospitalization and severe illness.” Currently MesenCure is only available to COVID-19 patients suffering from severe cases through compassionate use in Israel. Following the results of the phase II clinical trial, Bronshtein said Bonus BioGroup is now seeking emergency use approval starting in Israel. The company is also continuing to run phase III studies. (SD-Agencies) | 
